
The molecular geometry of CS2 can be determined by considering its Lewis structure and the VSEPR theory. 
Determination of the shape of CS2 molecule Therefore, CS2 does not have a positive or negative charge on either end of the molecule, making it non-polar. Additionally, the molecule has no lone pairs of electrons, which further contributes to its symmetry.Īs a result of this symmetry, the dipole moments of each carbon-sulfur bond cancel each other out, resulting in a net dipole moment of zero. The carbon-sulfur double bonds are non-polar since both atoms have the same electronegativity value. The CS2 molecule is non-polar due to its symmetrical linear shape and the equal sharing of electrons in its covalent bonds. Explanation of the polar/non-polar nature of CS2 molecule Double-check to ensure that each atom has a complete octet by counting the total number of valence electrons in the structure.īy following these steps, the CS2 Lewis structure can be drawn accurately.In CS2, there are no formal charges since each atom has satisfied the octet rule. Check the Lewis structure for formal charges, which are charges assigned to individual atoms based on the distribution of electrons in the molecule.In the case of CS2, all the valence electrons have been used up by forming double bonds between the carbon atom and each sulfur atom. If there are remaining electrons after forming single bonds, add them to the outer atoms as lone pairs until they satisfy the octet rule.Place two electrons between each atom to form single bonds, which will satisfy the octet rule for each atom.In CS2, the carbon atom is located in the center, with two sulfur atoms bonded to it on either side. Place the atoms in a linear arrangement to determine the bonding arrangement.For CS2, there are 16 valence electrons (4 from carbon and 6 from each sulfur). Calculate the total number of valence electrons in the molecule by adding the valence electrons of each atom.Carbon has 4 valence electrons, while each sulfur atom has 6 valence electrons. Determine the number of valence electrons for each atom in the molecule.Here are the step-by-step instructions to draw the CS2 Lewis structure in an active voice: In the case of CS2, there are two double bonds between the carbon atom and the sulfur atoms. This can be achieved by sharing electrons between the atoms to form covalent bonds. Then, the electrons are placed around the atoms to satisfy the octet rule, which states that each atom should have eight electrons in its valence shell. To draw the CS2 Lewis structure, one must first determine the number of valence electrons for each atom, which is 4 for carbon and 6 for sulfur. The CS2 Lewis structure is a representation of the arrangement of valence electrons in the molecule. 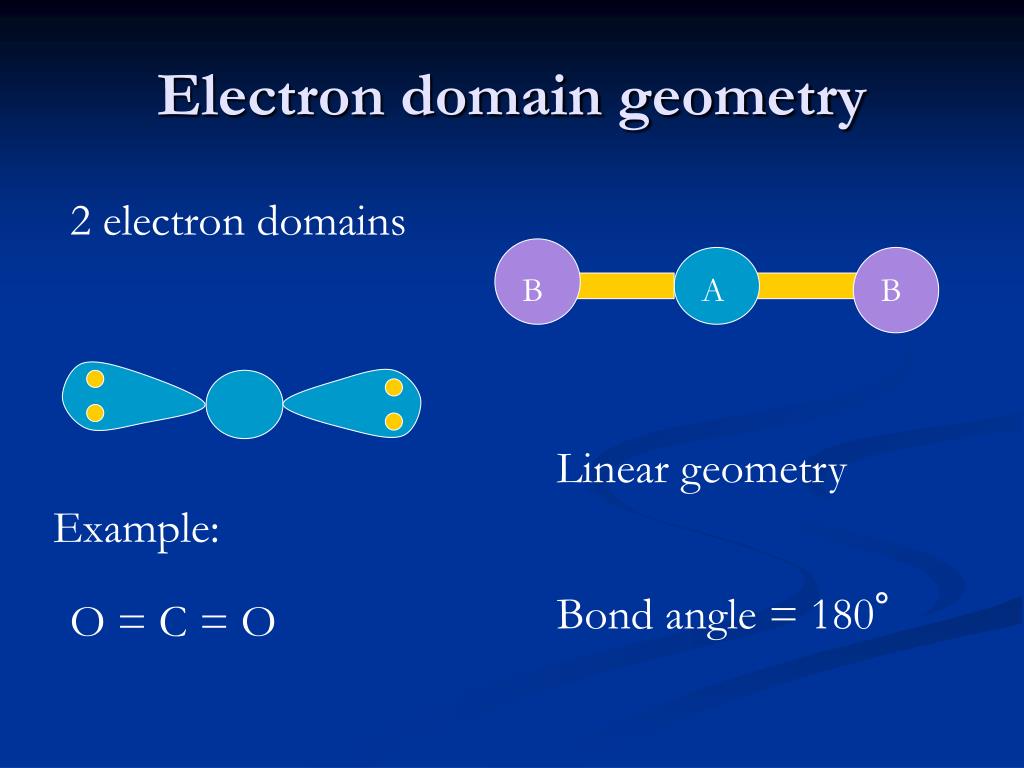
The CS2 Lewis structure is important for understanding the chemical properties and behavior of the molecule. Carbon disulfide is a highly flammable, colorless liquid with a pungent odor. It consists of one carbon atom and two sulfur atoms, bonded by covalent bonds. The chemical formula of Carbon disulfide is CS2.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
